Revista Ciencia e Ingeniería
Neogranadina
Sistema de Información Científica Redalyc
Red de Revistas Científicas de América
Latina y el Caribe, España y Portugal
Landfill leachate treatment by batch supercritical water oxidation
Tratamiento de
lixiviados de relleno sanitario por oxidación en agua
supercrítica
Victor Fernando Marulanda
Cardona *
vfmarulanda@lasalle.edu.co
Universidad La Salle ,
Colombia
Paola Andrea Marulanda Buitrago **
pmarulanda66@unisalle.edu.co
Universidad La Salle ,
Colombia
Didier Haid Alvarado Acosta ***
didier.alvarado- acosta.2016@uni.strath.ac.uk
Universidad de Strathclyde , Reino Unido
Tratamiento
de lixiviado de vertedero por lote de oxidación supercrítica de
agua
Ciencia e Ingeniería Neogranadina , vol. 27 , no. 2 , 2017
Universidad
Militar Nueva Granada
Disponible en: http://www.redalyc.org/articulo.oa?id=91150559003
Recibido: 22 de septiembre de 2016
Aceptado: 12 de mayo de 2017
Cómo citar: V. F. Marulanda Cardona, P. A. Marulanda Buitrago y D. H.
Alvarado Acosta, “Landfill Leachate
Treat-ment by batch supercritical water oxidation,” Ciencia e
Ingeniería Neogranadina, vol. 27, no. 2, pp. 5-26, 2017. DOI: http://dx.doi.org/10.18359/rcin.2305
Abstract: Landfill leachate treatment has been the
focus of a great deal of research through different physicochemical and
biological methods. However, no single method successfully addresses the
required destruction efficiencies regarding organic matter and nitrogen, which
is why the treatment is done employing combined technologies. Supercritical
water oxidation (SCWO), a process that takes place at temperatures and
pressures above the critical point of water and in the presence of a source of
oxygen, has been successfully applied to the treatment of different types of
wastewaters in an efficient way. Therefore, this paper presents an experimental
study of the supercritical water oxidation of landfill leachate in a batch
reactor in the temperature range 400-500°C, reaction times from 15 to 30
minutes and oxygen excess (OE) from 100% to 300 %. Total organic carbon
(TOC) and Total nitrogen (TN) destruction efficiencies were measured in the
reactor effluent samples and the combined effect of the studied factors was
analyzed by means of the Analysis of Variance (ANOVA). Optimal operation
conditions for TOC destruction were 400°C, 30 min and 100 % OE, being
500°C, 30 min and 100 % OE for TN destruction. Contrary to what has
been reported in similar studies, the results suggest that it is possible to
accomplish the simultaneous TOC and TN destruction in leachate wastewater by
SCWO treatment at 400°C, 100 % OE and residence times longer than 30
min and without using a catalyst, either in batch or in a continuous process,
as long as both the oxidant and the wastewater are mixed and heated together at
the reaction temperature.
Keywords:
Landfill leachate, SCWO, TOC destruction, Total
Nitrogen destruction.
Resumen: El tratamiento de lixiviados por medio de diferentes procesos fisicoquímicos
y biológicos ha sido ampliamente estudiado. Sin embargo, ningún
proceso logra las eficiencias de destruc- ción requeridas en cuanto a materia orgánica
y nitrógeno, razón por la cual el tratamiento se realiza por
medio de tecnologías combinadas. La oxidación en agua supercrítica
o SCWO, proceso que se lleva a cabo a temperaturas y presiones superiores a las
del punto crítico del agua en presencia de una fuente de oxígeno,
se ha aplicado exitosamente al tratamiento de distintos tipos de aguas
residuales de forma eficiente. Por lo tanto, este trabajo presenta un estudio
experimental de la oxidación en agua supercrítica de lixiviados
de relleno sanitario en un reactor batch, en el rango
de temperatura de 400-500°C, tiempos de reacción de 15 a 30 mi- nutos y excesos de oxígeno (OE) de 100 % a 300 %. Se
midieron las eficiencias de destrucción de carbono orgánico total
(COT) y nitrógeno total (NT), y se determinó el efecto combinado
de los factores estudiados por medio del Análisis de Varianza (ANOVA).
Las condiciones de operación óptimas para la destrucción
de COT fueron 400°C, 30 min y 100 % OE, y 500°C, 30 min y 100 % OE para
el NT. A diferencia de lo reportado en estudios similares, los resultados su- gieren que es posible llevar a cabo la destrucción
simultánea del COT y el NT en los lixiviados por medio de SCWO a
400°C, 100 % OE y tiempos de residencia de más de 30 min sin usar un
catalizador, ya sea en un proceso batch o continuo,
siempre y cuando tanto el oxidante como el agua residual se mezclen y se calienten
juntos a la temperatura de reacción.
Palabras clave: Lixiviados, SCWO, destrucción
de COT, destrucción de nitrógeno total.
Introduction
Municipal solid waste
management dumped inappropriately in nonengineered landfill
sites generates highly contaminated wastewater called leachate [1]. Landfill leachate
results from the degradation of the waste organic fraction in combination with
percolation of rainwater through solid waste piles [2]. Due to different biochemical
processes that take place as a result of the stabilization of the organic
matter in the waste pile, landfill leachate is heavily contaminated with
organic matter and ammonia nitrogen from the ammonification of organic nitrogen,
heavy metals and other toxic and priority pollutants. Environmental impacts
caused by wastewater of such characteristics, associated with the depletion of
oxygen and eutrophication in receiving surface and ground waters, are well
documented. Management of landfills could result in several environ-mental
impacts, for example, the percolation of leachates into the groundwater [3].
There-fore, an efficient wastewater treatment for landfill leachate must abate
not only the organic matter load but also nitrogen and heavy metals,
irrespective of the variations in volumetric flow and composition. According to
the International Solid Waste Association, a landfill is the “engineered
deposit of waste onto or into land in such a way that pollution or harm to the
environment is prevented” [4].
In this
aspect, landfill leachate treatment continues to be an area of intense research
by different physicochemical, biological and emerging methods. However, no
single technology claims to handle this heavily polluted residue in an
efficient way or compliance with the most stringent regulations without operational
constrains that limit in one way or another its practical application. Several
authors have extensively reviewed the ad-vantages and drawbacks of conventional
as well as emerging leachate treatment process using leachate transfer,
biodegradation, physical and chemical methods and membrane processes [2], [5]. It is evident from these
thorough reviews that the application of the conventional physicochemical and
biological treatment technologies to leachate treatment is hindered not only by
insufficient chemical oxygen demand (COD) and nitrogen removal efficiencies,
but also for operational constraints such as the age of the leachate in the
case of anaerobic biological processes, excessive sludge production, ammonia
inhibition and energy expenditure in aerobic biological processes, and sludge
production and chemicals products consumption in the case of physicochemical
processes. With only a few exceptions, COD removal efficiencies varied from
poor to moderate, and ammonia nitrogen, recalcitrant compound and considered
the rate-limiting step in the total mineralization of organic matter [6], [7], is scarcely addressed. For
example, reverse osmosis technology has complemented or replaced conventional
landfill leachate treatment. The large volume of concentrate needs to be
properly treated. Labiadh et al. [8] used anodic oxidation
combined with electro-Fenton processes to treat the concentrate and found,
under the most favorable electro-Fenton conditions, removal rates of 60 %
and 22 % for COD and TN, respectively. Xu et al. [9] also used a Fenton process
for leachate treatment membrane concentrates and reached 68.9 % TOC
reduction. No reference was made to TN or ammonia nitrogen reduction in this
study.
Also, and
in spite of all of the experimental work reported, it is not possible to make
general recommendations regarding the operational conditions and expected performances
of conventional processes. For example, hydraulic retention times (HRT) for
activated sludge processes, sequencing batch reactors and digesters, vary from
a few hours to several weeks for treatment of leachate with similar COD [5]. More recently, membranes, especially reverse osmosis (RO), have emerged
as an alternative to physicochemical and biological processes. Issues related
to fast membrane fouling and the necessary chemical cleaning procedure, which
shortens lifetime and decreases the productivity of the membrane, remain as
serious drawbacks. A physicochemical process as a pretreatment step before RO
could improve the operation while reducing the frequency of membrane cleaning
and consumption of chemicals [10].
A great
deal of potential impact can be associated with ammonia nitrogen and heavy
metals in the environment. Therefore, an all-around leachate treatment process
should be assessed not only from the organic matter removal, measured as COD,
BOD or TOC reduction, but also ammonia nitrogen and heavy metals abatement. At
the same time, it must be done through a single process system, not easily
affected by variations in waste chemical composition, nor requiring any additional
pretreatment operations, such as pH conditioning or ammonia stripping. Lastly,
it should not produce an additional byproduct to be further dealt with, such as
biological sludge and gases, as occurs in biological treatment processes [2], [5]. All of these criteria could
be satisfied by a process known as supercritical water oxidation or SCWO. This
is an advanced oxidation process that takes advantage of the fascinating
properties of supercritical water as a reaction medium at conditions of
temperature and pressure higher than the critical point of water (374 °C
and 22.1 MPa) [11]. At the-se conditions, and
as a result of the loss of hydrogen bonding, water is completely miscible with
organic compounds and oxygen. Therefore, it is possible to carry out rapid
oxidation reactions in a single-phase media at very high temperature without
mass transfer limitations. Hydrogen peroxide solutions, pure oxygen and
compressed and enriched air are used as oxidants in SCWO. Although the required
extreme pressure and temperature conditions could impose a high-energy
expenditure, SCWO has the potential to be considered a clean energy process [12]. The heat released by the
oxidation reaction could be converted to heat and shaft work, assuring a
self-sustained reaction, as well as generate excess shaft power to drive both
the high-pressure pump and the air compressor, as shown by
García-Rodríguez [13]. Several studies have
performed supercritical processes economic assessments through process
simulation [14]. While these processes
require a higher capital investment due to the cost of high-pressure equipment,
in the long run, are more profitable due to the possibility of recovering
energy from the reactor outlet, cheaper feedstocks and the reduced number of
process operations [15], [16].
SCWO has
been successfully applied to the treatment of different kinds of persistent and
anthropogenic wastes characterized by a high COD and heavy metals
concentration. These wastes cannot be efficiently or economically handled by
conventional waste treatment technologies such as incineration. Cui et al. [17] reported a COD removal rate
up to 92 % on oily sludge wastes in less than 10 minutes of reaction time
(RT) at 450 °C, 25 MPa, and 427 % oxygen excess. Akg et al. [18] studied the treatment of
textile wastewater by SCWO and reported a decrease in the chemical oxygen
demand up to 98.52 % in short reaction times, between 4-12 s and
temperatures between 400-600 °C at 25 MPa. The difference in reaction times
was attributed to the reaction system, being shorter in the continuous one.
Du et al. [19] reported a continuous flow
supercritical water oxidation process to treat high strength COD co-king
wastewater, which also contained high concentrations of NH3–N, phenol,
and inorganic salts. COD destruction close to 99 % at temperature
650°C, a pressure of 25 MPa, reaction time 24 s and 300 % excess
oxygen was achieved. Veriansyah et al. [20] reported the SCWO of
wastewater from liquid crystal display (LCD) manufacturing process, which
contains a wide variety of organic pollutants and chromium. A COD conversion
greater than 99.99 % was obtained at 615°C, 25 MPa, 998 %
oxygen excess and 10 s residence time and more than
97% chromium in the wastewater was recovered as chromium oxide. Marulanda and
Bolaños [15] studied the SCWO of a heavily PCB-contaminated trans-former oil in a continuos
process. It was achieved a 99.6 % organic matter conversion and
destruction of PCBs under the analytical technique detection limit at
539°C, 24.1 MPa, and 350% oxygen excess. Despite its advantages over
conventional end-of-pipe technologies, SCWO has met difficulties at a full
scale commercial activity, as reviewed by Marrone [21]. Corrosion, plugging,
materials durability and out-of-spec feed are the main reasons behind the
shutdown of several full -scale plants. New companies continue to enter the
field.
Leachate
treatment by supercritical water oxidation has been recently reported by several
groups using either continuous or batch processes. Gong and Duan
[22] studied a landfill leachate treatment process using a continuous system
with a transpiring-wall reactor (TWR). In this study, diluted leachate and
excess oxidant were fed to the reaction system. COD removal efficiencies were
higher than 97 % at 430 C and 30 MPa. Alternatively, Wang et al. [23] studied the supercritical
water oxidation of landfill leachate with a special focus on ammonia in a batch
reactor, at temperatures 380-500°C, reaction time of 50-300 s, oxygen
excess (OE) from 100 to 450 % and pressure of 25 MPa, with and without
MnO2 as a catalyst. Through a response surface analysis, it was
shown that NH3 and COD conversions were low at temperatures
near 400 °C and short reaction times. Zou et
al. [24] studied the co-destruction
of organic pollutants in leachates and dioxins in fly ash from leachates
incineration using super-critical water oxidation in a batch reactor at
different temperatures from 400-500°C, Oxy-gen excess from stoichiometric
to 300 % and residence times of 1 and 2 min. Similar conclusions to those
reported by Wang et al. [23], with a maximum COD
conversion of 99.2 % at 500°C, 300 % OE and 2 min and PCDD/F
degradation efficiency >90 %, were reported. Nitrogen was not
addressed in this study.
A leachate
treatment process based on SCWO technology could improve on existing physicochemical
and biodegradation processes in aspects such as the organic matter and
ammonia-nitrogen conversion, as well as the absence of sludge that needs
further treatment [21]. In order to assess the
feasibility of the scale-up of the process to an industrial level, additional
experiments should be conducted to elucidate the effect of the different
variables in the leachate treatment by SCWO, in such a way that the lab scale
experiments give insights on a possible pilot plant operation. Accordingly, the
batch SCWO of landfill leachate with hydrogen peroxide is reported in this
work. Process performance is assessed employing a factorial experimental
design. Individual factors and its interactions in Total Organic Carbon (TOC)
and Total Nitrogen (TN) destruction efficiencies were analyzed.
1. Materials and
Methods
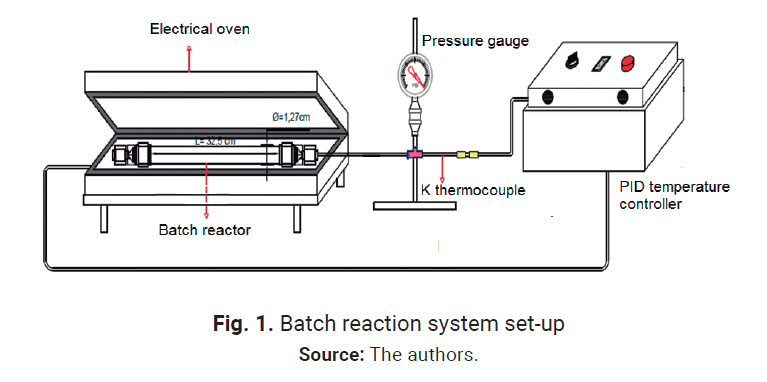
1.1. Experimental apparatus
SCWO experiments were
carried out in a batch reactor made of ½” (1.27 cm) OD Swagelok
tubing and fittings, 0.17 cm wall thickness, and 25 cm length for a total
reaction volume of 16.98 cm3. The reactor withstands a maximum
pressure of 35 MPa at 500 °C. A PID temperature
controller oven (± 2°C) was used to carry out the experiments.
1.2. Material and analytical
methods
Landfill leachate was
collected from a sanitary landfill located in the city of Villavicencio in
Colombia, in a WWTP affluent sampling port. HCl to pH
2 was added to preserve the sample and was protected from sunlight and
atmospheric oxygen. The sample was kept in a freezer. Hydrogen peroxide (H2O2)
30 wt % solution (Merck) was used as the
oxidant. Hydrogen peroxide decomposition in supercritical water produces OH
radical, which is extremely reactive. However, it is also very unstable and
rapidly decomposes to oxygen [25], [26].
Total organic carbon
(TOC) and total nitrogen (TN) were measured before and after SCWO treatment
following adapted methods corresponding to standardized methods 5310D for TOC
in a DR 3800 Hach Spectrophotometer,
and TNb 220 for organically and inorganically bound
nitrogen in a Macherey-Nagel nano
color Vario 4 Spectrophotometer. Instruments blank
were prepared with distilled and deionized water after adding the contents of
the ampuls or decomposition reagents required for
each method, digested in thermo reactor and set to 0.0 mg/L in both cases. TOC
is considered more convenient than BOD or COD due to the presence of organic
carbon that does not respond to either of these methods. Total Nitrogen is the
sum of nitrate-nitrogen (NO3-N), nitrite-nitrogen (NO2-N),
ammonia-nitrogen (NH3-N) and organically bonded nitro-gen. Several
authors have shown that nitrate and nitrite nitrogen can be formed in SCWO.
Therefore, TN is more representative of nitro-gen fate in SCW than Total Kjeldahl Nitrogen (TKN), which has into account only
ammonia and organic nitrogen.
Total organic carbon and
total nitrogen destruction efficiencies were defined according to Equations (1)
and (2):
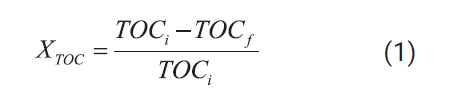
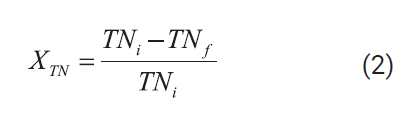
Where TOCi and TNi
are the initial concentration of total organic carbon and total nitrogen in the
leachate after the dilution with 30 wt% hydrogen
peroxide solution and TOCf and TNf are the corresponding concentrations in the
reactor effluent.
1.3. Experimental procedure
In a typical batch run,
a volume of leachate sample is added to the required volume of 30-wt% hydrogen
peroxide solutions to provide the oxygen according to the desired oxygen excess
for a specific run. Then an amount of reaction mixture is added to one end of
the reactor utilizing volumetric pipettes and sea-led with screw caps. The
reactor is placed in an electrical insulated clamp-type oven previously heated
to the desired reaction temperature, as shown in Fig. 1 and the batch re-action
time is set to zero. Once the reaction time is complete, the oven is open and
the reactor removed and rapidly quenched in a water bath to stop the reaction.
TOC, TN, and pH were
measured in the raw landfill leachate sample as 8700 mg/L, 2800 mg/L and 6.3,
respectively. In a typical experimental run, a given amount of undiluted
leachate was mixed with a volume of 30 wt% hydrogen
peroxide solution, according to the amount of oxidant excess and having into
account the reactions for hydrogen peroxide decomposition in SCW [26] and TOC
oxidation (Ecuations 3 and 4):
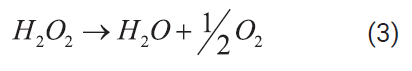
![]()
Accordingly, oxygen
excess (OE) was defined as in Equation (5):
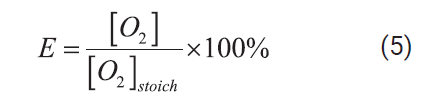
Where [O2]
is the provided initial concentration of O2 from the decomposition
of the added hydrogen peroxide according to the decomposition reaction and [O2]stoich is the
calculated stoichiometric amount of O2, according to Equation (4),
required to completely oxidize the organic matter measured as TOC, 23200 mg/L
of oxygen for 8700 mg/L TOC, which is provided by 49300 mg/L of hydrogen
peroxide, 164.3 g/L of 30 wt% hydrogen peroxide
solution. Calculated oxidant solution requirement based on this approximation
is considerably lower than that resulting from the most widely used COD
measurement. The exact mass of leachate-oxidant solution to be injected in the
reactor is determined based on the specific volume of water at re-action
conditions calculated by means of the Peng-Robinson
Equation of State (PR-EOS) and the volume of the reactor. PR EOS has been shown
to perform as well as the more complicated perturbed hard sphere Van der Waals
equations, with a good qualitative picture of all types of phase behavior and
reasonable quantitative representation for a variety of systems [27], [28]. The
reactor is placed in the oven, previously heated at reaction conditions, and
left for a specific reaction time. Once the reaction time is finished, the
reactor is removed from the oven and immersed in a cold water bath to stop the
reaction. Liquid effluent is collected for TOC and TN analysis. No attempt was
made to collect the gaseous phase at this stage of the research.
1.4. Statistical analysis
Experimental runs were
planned according to a replicated twice 2k full factorial design
with three factors (k): temperature (A), reaction time (B) and oxygen excess
(C), using TOC and TN as response variables. Low and high levels of each factor
(-1, +1) were selected according to preliminary experiments as well as
practical considerations regarding reactor construction materials and hydrogen
peroxide consumption. Thus, levels of temperature were 400 and 500 °C, 15
and 30 min reaction time and oxygen excess of 100 and 300 %. With four
repetitions in the central point (450 °C, 22.5 min, and 200 % OE),
the full design is comprised of 20 experimental runs, made in randomized order.
Statistical analysis of the results was made according to the analysis of
variance (ANOVA) on both response variables, as well as the graphical analysis
of the significant main effects and interactions plots, by using the
statistical software package Minitab.
2. Results and
discussion
The summary of reaction
conditions, initial and final TN and TOC concentrations and destruction
efficiencies, is shown in Table 1, in which T is the reaction temperature and
Time is the total reaction time. Runs are identified and shown according to the
label in the 2k experimental design and the order in which each run
was carried out. For exam-ple, run 5-bc means it was
the 5 run in the experimental design, with factor (A) in the level low,
reaction temperature 400°C, whereas factors B and C are in the level high,
re-action time and oxygen excess 30 min and 300 %, respectively. Run
8-(1) means all the factors were in the level low, 400°C, 15 min and
100 % OE.
For all the treatment
conditions in the experimental design, TOC destruction efficiencies were higher
than 70 %, whereas TN destruction efficiencies were lower, with efficiencies
higher than 30 %. Maximum destruction efficiency for TOC was 99.5 %
at 400°C, 30 min reaction time and 300 % OE, whereas for TN was 92.2 %
at 500°C, 30 min and 100 % OE. These results suggest the organic
matter in the leachate can be effectively oxidized at 400°C. However,
nitrogen abatement might require a more aggressive treatment at 500 °C. An
analysis of variance (ANOVA) of the experimental data was carried out in order
to elucidate the joint effect of the factors and its interactions on the response,
so that a more general conclusion regarding the optimal set of operation
conditions can be drawn. ANOVA for TOC and TN destruction efficiencies is
discussed next.
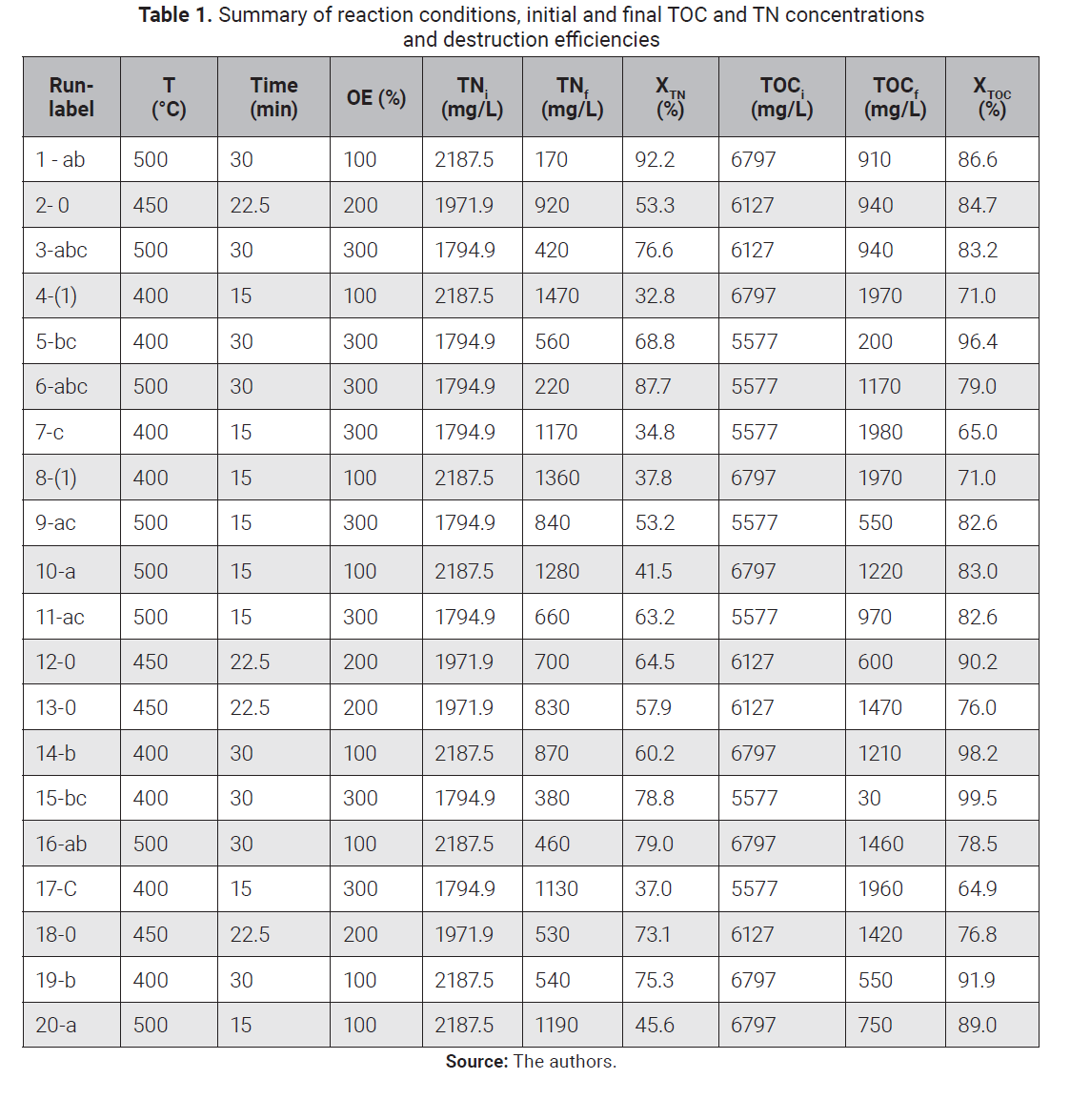
2.1 Total organic carbon ANOVA
According to the TOC
normal plot of standardized effects shown in Fig. 2, factor B (Reaction time)
and the interaction AB (Temperature and reaction time) have a significant
effect on the response. However, factor A (Temperature) and factor C (oxygen
excess) did not have a significant effect. Temperature and oxygen have a
positive effect on oxidation re-actions rates, and SCWO studies are usually
carried out in a continuous manner with a few seconds of residence time at
temperatures higher than 500°C and oxygen excess over 300 % [15],
[20]. The lack of a positive effect in both factors could be attributed to the pro-longed
reaction time considered in this study, which was long enough, even in the
level low, for the oxidation reaction to proceed almost
to completion. On the other hand, as expected in any reaction, the interaction between
temperature and time means working at higher temperature results in a shorter
reaction time. The nature of the interaction for this study is discussed next.
The model had a
regression coefficient R2= 88.39 %. Since A, C, BC, AC and ABC
terms are insignificant; the design was re-analyzed dropping these terms from
the model. The normal probability plot of the residuals and
residuals versus. fitted values or constant
variance plot for the re-analyzed design are shown in Figs. 3 and 4.
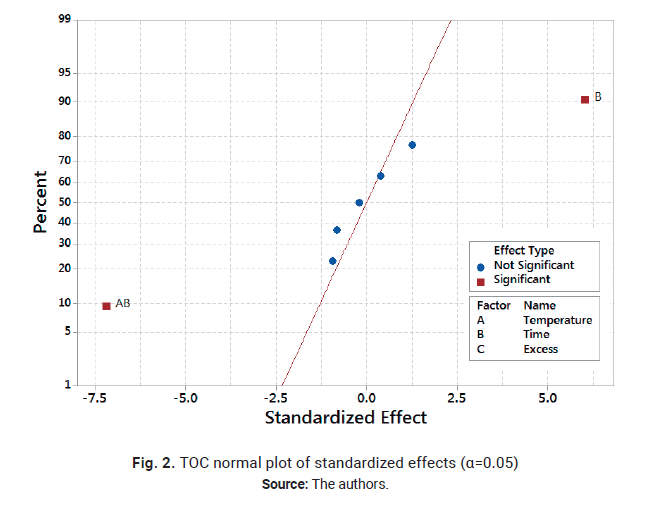
 .
.
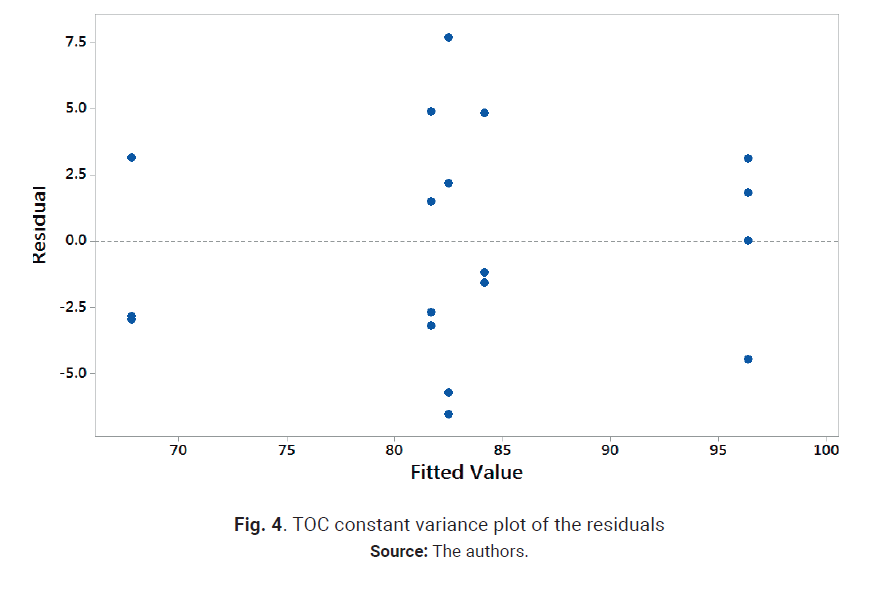
As can be observed in Fig. 3, the normal probability plot of the residuals resembles a straight
line; therefore the error distribution is normal. In a similar way, Fig. 4
shows a random patter of the residuals on both sides of 0, without any
recognizable patter-ns. Therefore, the model is correct and the assumptions
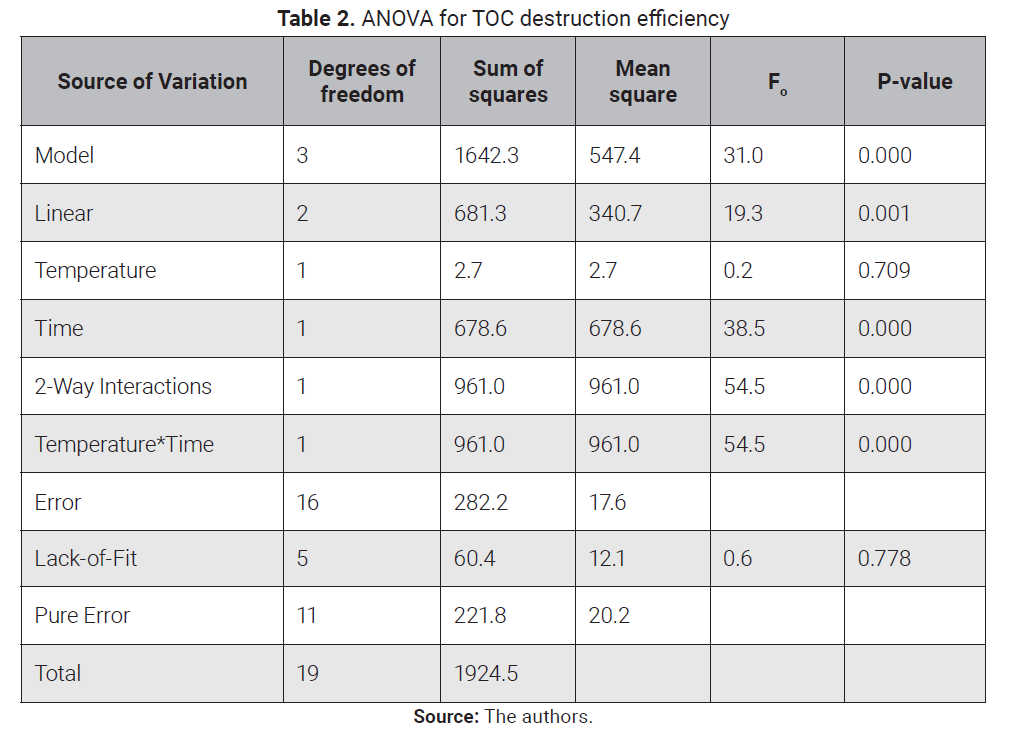
were satisfied. ANOVA for the re-analyzed model is summarized in Table 2 and
resulting model is given in Equation (6)
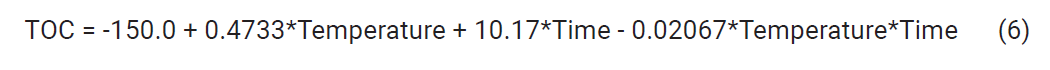
The standard deviation
was 4.19959. Accor-ding to the P-values in the ANOVA for TOC destruction
efficiency in Table 2, the model has a P-value of 0.000, whereas lack of fit was
not significant, which means the model fits well the data. In a similar way,
the main effects of reaction time (B) and the interaction AB of temperature and
reaction time are highly significant, with P-values < 0.01.
According to Montgomery
[29], the main effects do not have much meaning when there are involved
significant interactions. While the positive effect estimated for B factor
suggest working at the high level of the reaction time to increase the TOC
destruction efficiency, AB interaction might indicate the opposite trend. AB
interaction plot is shown in Fig. 5.
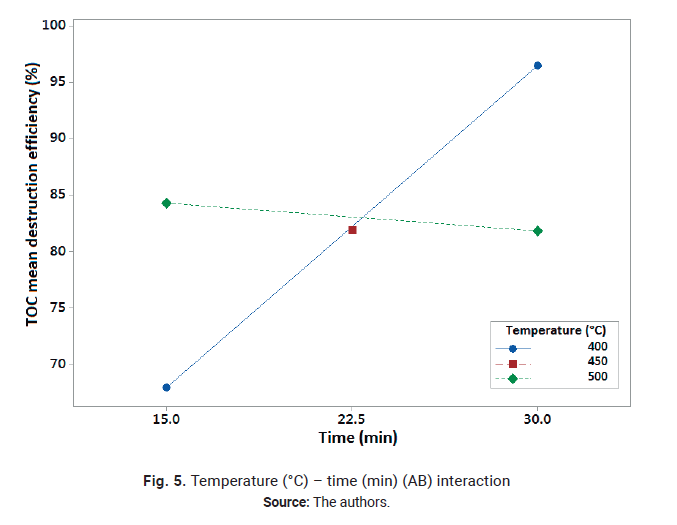
Fig. 5 shows that the effect
of time is significant when working at 400°C and negligible when working at
500°C. The higher reaction temperature speeds up the reaction and as a
result, the TOC mean destruction efficiencies are similar at 15 and 30 minutes,
as was previously discussed. However, TOC mean destruction efficiencies are
higher when working at 400°C and 30 min reaction time. Usually, SCWO
experimental studies report a positive effect of temperature when working in a
continuous unit set up and with residence times of a few seconds [15], [30].
The heating time of the substance to oxidize and the oxidant agent pumped separately,
is not accounted for as part of the total reaction time and could be even
longer than the residence time in the reactor. From Fig. 5 it can be concluded
that the optimal operation conditions for TOC destruction are 400 °C, 30
min reaction time and 100 % oxygen excess since this factor was not significant.
However, factors effect on TN destruction could be different and suggest a
different set of operation conditions. TN ANOVA is discussed next.
2.2 Total nitrogen ANOVA
ANOVA for TN
destruction efficiency is shown in Table 3, and TN normal plot of standardized
effects is presented in Fig. 6. Data standard deviation was 7.18584. The model
in Equation (7)
had a P-value of 0.000, and an R2 value of 90.23 %, lack of fit was not significant,
which means the model fits well the data. Main factors A and B, temperature and
time, have a significant effect on nitro-gen abatement, with P-values for
temperature and time of 0.001 and 0.000, respectively. Oxidant excess did not
have a positive effect, which can be attributed again to the prolonged reaction
time. Differently, from what was observed for TOC destruction, there are no
significant interactions for TN. Accordingly, the design was re-analyzed
dropping the terms C, AC, BC, AB and ABC from the analysis. The results are
shown in Figs. 7 and 8. Nor-mal probability plot resembles a straight line, and
residuals show a random pattern around 0; therefore, there is no reason to
suspect any problems with the validity of the conclusion. TN temperature and
time main effects plot is shown in Fig. 9.
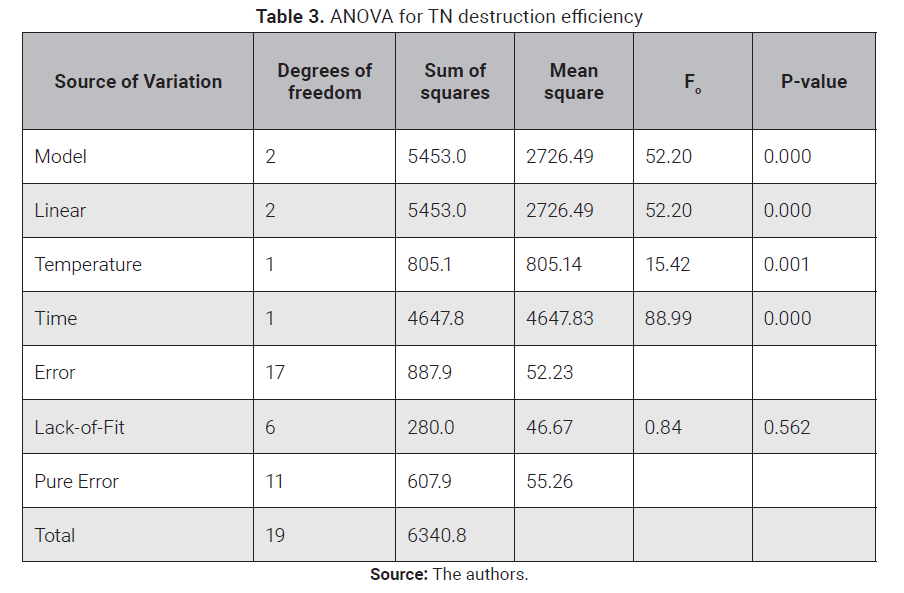
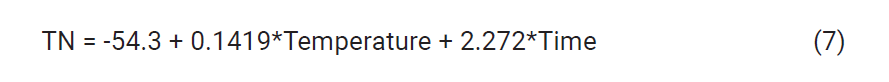
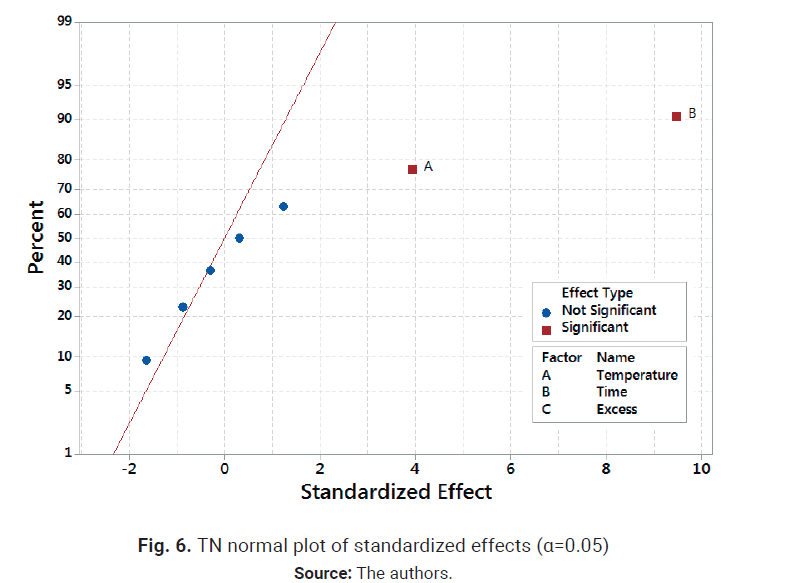
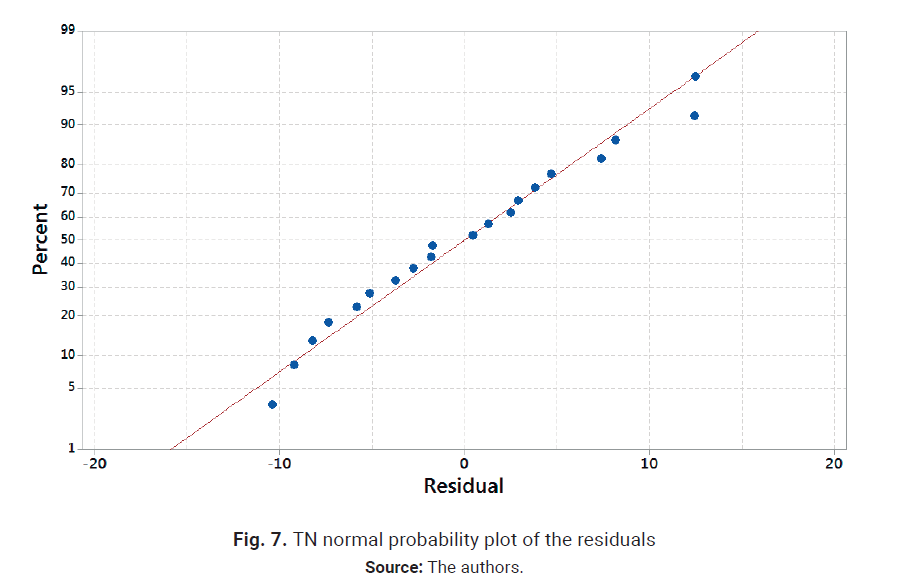
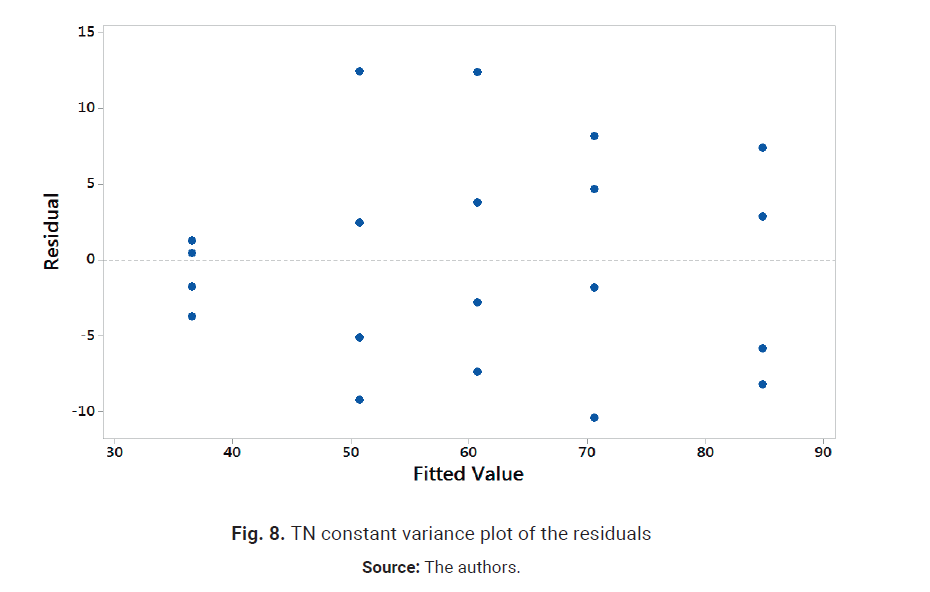
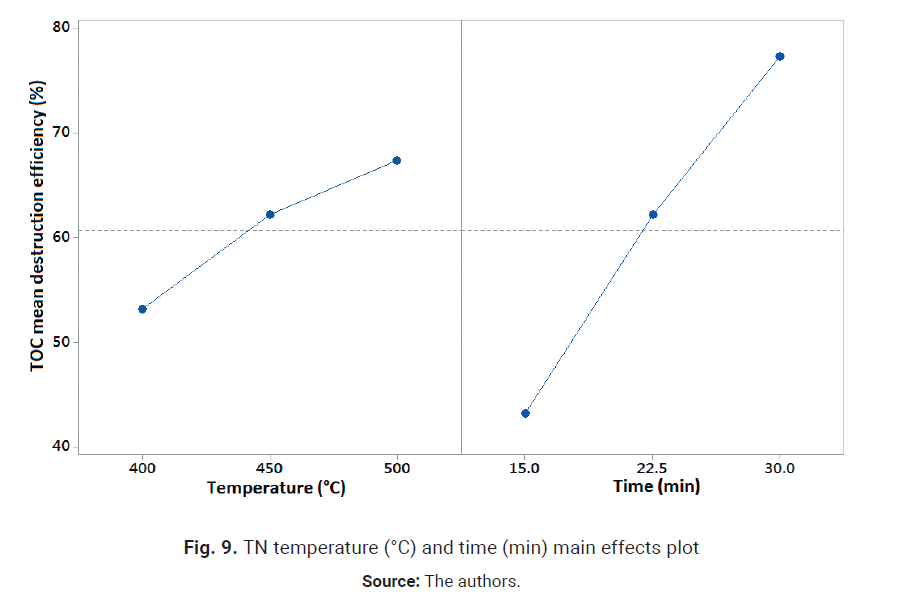
Fig. 9 shows the positive
effects of temperature and time, which suggest the optimal operation conditions
for achieving a high TN destruction efficiency are 500 °C, 30 min reaction
time and 100 % oxygen excess. Al-though temperature effect was found
insignificant for TOC destruction when working at residence times longer than
15 min, ammonia-nitrogen (NH3-N), formed as a stable intermediate compound in
the oxidation of nitrogen-containing components, is a well-known recalcitrant
compound and usually requires temperatures higher than 600 °C in the SCWO
process [6], [31], [32], [33]. In fact, it has been suggested the ammonia
oxidation to nitrogen or nitrous oxide is the rate limiting step in the global
oxidation [7]. At 400°C, 30 min and OE of 100 %, optimal operation
conditions for TOC destruction, average TN destruction of runs 14 and 19 is
68 %, which might be insufficient to meet the most stringent TN discharge
limits. However, this result is much better than the 46.9 % ammonia
conversion reported by Wang et al. [23] in the SCWO of leachate at 400°C,
OE of 350 % and 300 s reaction time, with a heating velocity of 4.07 °C/min
in the absence of the oxidant.
The difference could be
attributed to the experimental procedure followed in this work, in which the leachate
and oxidant were mixed and heated up to reaction conditions and left for a
particular time. In this aspect, hydrogen peroxide rapidly decomposes with
temperature, producing reactive OH free radicals [26], which could promote fast
organic matter oxidation reactions. Heating up separately could cause pyrolysis
of the organic compounds [34], [35] as well as the thorough decomposition of
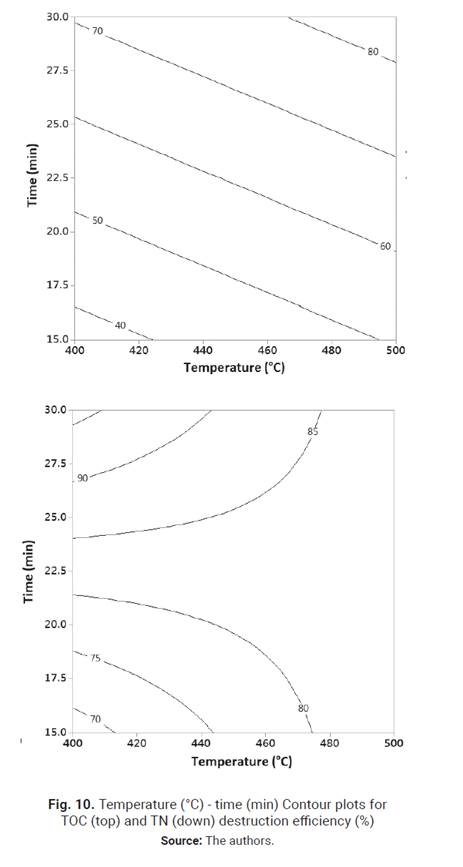
peroxide to oxy-gen, which is a less reactive oxidant agent. Temperature-time
contour plots for TOC and TN destruction efficiencies, generated by the
statistical software Minitab from data quadratic regression models in the
studied experimental region, are shown in Fig. 10. As it was shown in TOC
ANOVA, the temperature-time significant interaction causes the TOC destruction
efficiency contour plots to curve when temperature goes from low (400°C) to
high (500°C), whereas TN con-tour plots are straight due to the absence of
significant interactions and TN destruction efficiency increases with the
temperature and time. Although high TN destruction efficiency can be obtained
at 500°C, as shown in TN contour plots and also suggested by other works
[23], Fig. 9 indicates that a reaction time longer than 30 min could increase
TN destruction efficiency. Therefore, not only high TOC but also TN destruction
efficiency could be obtained working at 400°C, OE of 100 % and
reaction times longer than 30 minutes. From a scale-up point of view, a
reaction temperature of 400°C is more desirable than 500°C due to
increased cost of equipment construction materials and also the higher energy
consumption expected when working at 500°C. Even though the results were
obtained in a batch reactor, the implemented experimental procedure of mixing
the leachate and oxidant and heating up to reaction conditions would be similar
to the operation of a continuous unit in which the leachate-oxidant mixture is
pumped through preheaters up to the reaction temperature and kept constant for
a residence time similar to those found in this study.
3. Conclusions
This work reports the
SCWO of landfill leachate in a batch reactor system in which the joint effect
of temperature, time and oxygen excess were assessed using a factorial
experimental design. ANOVA for TOC and TN destruction efficiency showed the
temperature-time interaction was significant for TOC destruction efficiency and
the interaction plot showed the optimal operation conditions for TOC
destruction were 400°C, 30 min and 100 % OE. Temperature and time
factors were significant for TN destruction efficiency, which were higher than
those reported by other works at similar reaction conditions: main effect plots
showed the optimal operation conditions were 500°C, 30 min and 100 %
OE. Although the higher temperature is required for TN destruction, contour
plots showed an even longer reaction time could increase TN destruction
efficiencies while keeping the reaction temperature at 400°C, a more
desirable reaction temperature from a scale-up point of view.
Finally, it is
important to strengthen the development of this type of research, mainly
because in the past some knowledge limitations have been found and one of the
biggest gaps is the easy way methodology to carry out a risk assessment,
specifically for land-fill leachate [36]. Likewise in different countries,
there is some evidence that shows the research on sources contaminated with
leachate was useful for enforcing the environ-mental regulations and improving
the environmental quality [37].
Acknowledgements
The authors wish to thank the Colombian
Institute of Science and Technology (COL-CIENCIAS) for a young researcher grand
awarded to engineer Paola Marulanda to carry out
research at Universidad de La Salle in Bogotá for one year.
[1] P. Ghosh,
I. S. Thakur and A. Kaushik, “Bioassays for
toxicological risk assessment of landfill leachate: A re-view,” Ecotoxicol.
Environ. Saf., vol.141,
pp. 259-270, Jan. 2017. https://doi.org/10.1016/j.ecoenv.2017.03.023
[2] T. A. Kurniawan, W. H. Lo and G. Y. S. Chan, “Physico-chemical treatments for removal of recalcitrant
contaminants from landfill leachate,” Jour. Hazard. Mater., vol.
129, no. 1-3, pp. 80-100, 2006. https://doi.org/10.1016/j.jhazmat.2005.08.010
[3] Y. N. Vodyanitskii, “Biochemical processes in soil and groundwater
contaminated by leachates from municipal landfills (Mini Review),” Ann. Agrar. Sci., vol.14, no. 3, pp. 1512-1887, 2016. https://doi.org/10.1016/j.aasci.2016.07.009
[4] N. B. Yenigül, A. M. M. Elfeki, J.
C. Gehrels, C. van den Akker,
A. T. Hensber-en and F. M. Dekking,
“Reliability assessment of groundwater monitoring networks at landfill
sites,” Jour. Hydrol.,
vol. 308, no. 1-4, pp. 1-17, 2005. https://doi.org/10.1016/j.jhydrol.2004.10.017
[5] S. Renou,
J. G. Givaudan, S. Poulain,
F. Dirassouyan and P. Moulin, “Landfill leachate
treatment: Review and oppor-tunity,” Jour. Hazard.
Mater., vol. 150, no. 3, pp. 468-493, 2008.
https://doi.org/10.1016/j.jhazmat.2007.09.077
[6] J. M. Ploeger, A. C. Madlinger and J.
W. Tester, “Revised Global Kinetic Measurements of Ammonia Oxidation in
Supercritical Water,” Ind. Eng. Chem. Res.,
vol. 45, no. 20, pp. 6842-6845, 2006. https://doi.org/10.1021/ie0605276
[7] M. D. Bermejo, F. Cantero and M. J. Cocero, “Supercritical
water oxidation of feeds with high ammonia concentrations Pilot plant
experimental results and modeling,” Chem. Eng. Jour.,
vol.137, no. 3, pp. 542-549, 2008. https://doi.org/10.1016/j.cej.2007.05.010
[8] L. Labiadh,
A. Fernandes, L. Ciríaco,
M.J. Pacheco et al., “Electrochemical treatment of concentrate from
reverse osmosis of sanitary landfill leachate,” J.
Environ. Manage.,
vol. 181, pp. 515-521, Oct. 2016. https://doi.org/10.1016/j.jenvman.2016.06.069
[9] J. Xu,
Y. Long, D. Shen, H. Feng and
T. Chen, “Optimization of Fenton treatment process for degradation of
refractory organics in pre-coagulated leachate membrane concentrates,” Jour. Hazard. Mater., vol. 323, pp. 674-680, 2017. https://doi.org/10.1016/j.jhazmat.2016.10.031
[10] S. Renou,
S. Poulain, J. G. Givaudan and
P. Moulin, “Treatment process adapted to stabilized leachates: Lime
precipitation-prefiltration-reverse osmosis,” Jour. Memb. Sci., vol. 313, no. 1-2, pp. 9-22, 2008.
https://doi.org/10.1016/j.memsci.2007.11.023
[11] J. R. Portela, E. Nebot and E. Mart, “Hydrothermal
oxidation: Application to the treatment of different cutting fluid
wastes,” Jour. Hazard. Mater., vol. 144, no. 3, pp. 639-644,
2007. https://doi.org/10.1016/j.jhazmat.2007.01.088
[12] M. J. Cocero, E. Alonso and M. T. Sanz,
“Supercritical water oxidation process under energetically
self-sufficient operation,”. Jour. Supercrit. Fluids, vol. 24, no. 1, pp. 37-46, 2002.
https://doi.org/10.1016/s0896-8446(02)00011-6
[13] Y. García-Rodríguez,
F. Mato, A. Martín, M. D. Bermejo and M. J. Cocero, “Energy recovery from effluents of
supercritical water oxidation reactors,” Jour. Supercrit. Fluids, vol. 104, pp. 1-9, Sep. 2015. https://doi.org/10.1016/j.supflu.2015.05.014
[14] V. F. Marulanda, “Biodiesel production by supercritical
methanol transesterification: Process simulation and
potential environmental impact assessment,” Jour.
Clean. Prod., vol. 33, pp.
109-116, 2015. https://doi.org/10.1016/j.jcle-pro.2012.04.022
[15] V. Marulanda
and G. Bolaños, “Super-critical water
oxidation of a heavily PCB-contaminated mineral transformer oil:
Laboratory-scale data and economic assessment,” Jour.
Supercrit. Fluids, vol. 54, pp. 258-265, Aug.
2010. https://doi.org/10.1016/j.supflu.2010.04.008
[16] J. M. N. van Kasteren and a. P. Nisworo, “A
process model to estimate the cost of industrial scale biodiesel production
from waste cooking oil by supercritical transesterification,”
Resour. Conserv. Recycl., vol. 50,
no. 4, pp. 442-458, 2007. https://doi.org/10.1016/j.resconrec.2006.07.005
[17] B. Cui, F. Cui, G. Jing, S. Xu et al., “Oxidation of oily sludge in supercritical
water,” Jour. Hazard. Mater., vol. 165, no. 1-3, pp. 511-517, 2009. https://doi.org/10.1016/j.jhazmat.2008.10.008
[18] M. Akg and O. Onur, “Treatment of textile wastewater by SCWO in a
tube reactor,” Jour. Supercrit. Fluids,
vol. 43, no.1, pp. 106-111, 2007.
[19] X. Du, R. Zhang, Z. Gan and J. Bi, “Treatment of high strength coking
wastewater by supercritical water oxidation,” Fuel,
vol. 104, pp. 77-82, Feb. 2013. https://doi.org/10.1016/j.fuel.2010.09.018
[20] B. Veriansyah,
T. Park, J. Lim and Y. Lee, “Supercritical water oxidation of wastewater
from LCD manufacturing process: kinetic and formation of chromium oxide
nanoparticles,” Jour. Supercrit.
Fluids, vol. 34, no. 1, pp.
51-61, 2005. https://doi.org/10.1016/j.supflu.2004.10.001
[21] P. A. Marrone, “Supercritical water oxidation. Current status of full-scale commercial activity for waste
destruction,” Jour. Supercrit. Fluids, vol. 79, pp. 283-288, Jul. 2013. https://doi.org/10.1016/j.supflu.2012.12.020
[22] W. Gong and X. Duan, “Degradation of landfill leachate using transpiring-wall
supercritical water oxidation (SCWO) reactor,” Waste
Manag., vol. 30, no. 11, pp. 2103-2107, 2010. https://doi.org/10.1016/j.wasman.2010.04.028
[23] S. Wang, Y. Guo, C. Chen, J. Zhang et al., “Supercritical water oxidation
of landfill leachate,” Waste Manag.,
vol. 31, no. 9-10, pp. 2027-2035, 2011. https://doi.org/10.1016/j.wasman.2011.05.006
[24] D. Zou,
Y. Chi, C. Fu, J. Dong, “Co-destruction of organic pollutants in
municipal solid waste leachate and dioxins in fly ash under supercritical water
using H2O2 as oxidant,” Jour. Hazard. Mater., vol.
248-249, pp. 177-184, 2013. https://doi.org/10.1016/j.jhazmat.2013.01.005
[25] K. Hatakeda,
Y. Ikushima, O. Sato, T. Aizawa
and N. Saito, “Supercritical water oxidation of polychlorinated biphenyls
using hydrogen peroxide,” vol. 54, no. 15-16, pp. 3079-3084, 1999. https://doi.org/10.1016/s0009-2509(98)00392-3
[26] E. Croiset,
S. F. Rice and R. G. Hanush, “Hydrogen peroxide
decomposition in supercritical water,” Am. Inst. Chem.
Eng., vol. 43, no. 9, pp. 2343-2352, 1997. https://doi.org/10.1002/aic.690430919
[27] M. Mukhopadhyay.
Natural Extracts Using Supercritical Carbon Dioxide.
Florida, USA: CRC Press, 2000. https://doi.org/10.1201/9781420041699
[28] J. L. Dinaro, P. A. Marrone, S. F.
Rice, P. A. Webley, “Critical review of kinetic
data for the oxidation of methanol in supercritical water,” Jour. Supercrit. Fluids, vol. 34, no. 3, pp. 249-286, 2005.
https://doi.org/10.1016/j.supflu.2003.12.018
[29] D. C. Montgomery. Design and Analysis of Experiments (8th.
Ed.). Nueva York: John Wiley & Sons, 2012.
[30] B. Veriansyah,
J. Kim and J. Lee, “Destruction of chemical agent simulants in a
supercritical water oxidation bench-scale reactor,” Jour.
Hazard. Mater., vol. 147, no. 1-2, pp. 13-19, 2007. https://doi.org/10.1016/j.jhaz-mat.2006.12.040
[31] R. Killilea,
K. C. Swallow and G. T. Hong, “The Fate of Nitrogen in
Supercritical-Water Oxidation,” Jour. Supercrit. Fluids, vol. 5, no.
1, pp. 72-78, 1992. https://doi.org/10.1016/0896-8446(92)90044-k
[32] G. Knothe,
A. C. Matheaus and T. W. Ryan, “Cetane numbers of branched and straight-chain fatty esters
determined in an ignition quality tester,” Fuel,
vol. 82, no. 8, pp. 971-975, 2003. https://doi.org/10.1016/s0016-2361(02)00382-4
[33] K. M. Benjamin and P.
E. Savage, “Supercritical Water Oxidation of Methylamine,” Ind. Eng. Chem. Res., vol. 44, no. 14, pp. 5318-5324,
2005. https://doi.org/10.1021/ie0491793
[34] S. Yesodharan,
“Supercritical water oxidation: An environmentally safe method for the
disposal of organic wastes,” Current Science,
vol. 82, no. 9, pp. 1112-1122, 2002.
[35] Z. Fang, S. K. Xu, R. L. Smith Jr., K. Arai and J. A. Kozinski,
“Destruction of deca-chlorobiphenyl in
supercritical water under oxidizing conditions with and without Na2CO3,” Jour. Supercrit. Fluids, vol. 33, no. 3, pp. 247-258, 2005.
https://doi.org/10.1016/j.supflu.2004.08.010
[36] T. E. Butt, E. Lockley and
K. O. K. Oduyemi, “Risk assessment of landfill
disposal sites - State of the art,” Waste Manag., vol. 28, no. 6, pp. 952-964, 2008. https://doi.org/10.1016/j.was-man.2007.05.012
[37] J. A. Reyes-López, J. Ramírez-Hernández,
O. Lázaro-Mancilla, C. Carreón-Diazconti
and M. M. L. Garrido, “Assessment of
groundwater contamination by landfill leachate: A case in México,”
Waste Manag., vol. 28,
no. 1, pp. 33-39, 2008. https://doi.org/10.1016/j.wasman.2008.03.024
Author notes
* Chemical Engineer, Ph.D. in Engineering.
La Salle University, Titular Professor.
Bogotá, Colombia. E-mail: vfmarulanda@lasalle.edu.co.
ORCID: http://orcid.org/0000-0001-8038-2536
** Environmental and Sanitary Engineer. Bogota,
Colombia. E-mail: pmarulanda66@unisalle.edu.co. ORCID:
http://orcid.org/0000-0001-9168-5591
*** Environmental and Sanitary Engineer, Student
of MSc. Hydrogeology, University of Strathclyde, Glasgow,
United Kindom. E-mail:
didier.alvarado-acosta.2016@uni.strath.ac.uk. ORCID:
http://orcid.org/0000-0002-7092-0721
HTML
generated from XML Redalyc JATS4R